WRAP Certification – Passport for Textile and Garment Enterprises in the Time of Integration
In the article below we will provide some information about the world’s largest independent certification program focusing on the field of apparel, footwear and sewing products (WRAP).
1. What is WRAP?
WRAP stands for Worldwide Responsible Accredited Production – the name of the organization that recognizes social responsibility in global production, focusing on the fields of apparel, footwear and apparel products.
The WRAP program evaluates compliance with WRAP’s 12 principles to ensure safe, legal and ethical manufacturing processes.
Include:
Comply with applicable laws and regulations
Prohibit the use of forced labor
Prohibit the use of child labor
Harassment and abuse are prohibited
Salary and benefits
Work time
Discrimination is prohibited
Occupational safety and hygiene
Freedom of association and collective bargaining
Environment
Comply with tariff regulations
Security
2. Benefits of Applying for WRAP Certification and Being Certified:
WRAP certification will affirm the commitment of the top leadership of the unit and organization to apply, maintain and regularly improve the occupational health and safety management system of workers to ensure Workers have the right to work in a safe environment, ensuring necessary and health conditions to improve the quality of work to meet requirements.
WRAP certification is evidence that helps an organization meet the legal requirements for responsibility requirements with production workers, who are low status and vulnerable in society, protected.
Manufacturers that obtain WRAP certification will help enhance their image, corporate culture and brand.
Improve product and service competitiveness as a condition for providing. Many businesses that receive WRAP training will work more productively and bring greater efficiency.
WRAP certification is a passport for goods that can be exported to European regions: USA, UK…. And developed countries prioritize human rights first
3. Three levels of WRAP certification:
There are three levels of WRAP certification – Platinum, Gold and Silver.
Certification is issued to a facility as determined by WRAP and depends on the extent to which an audit demonstrates full compliance and management commitment to the WRAP Principles.
Platinum
Platinum certification is awarded to facilities that have demonstrated full compliance with WRAP’s 12 Principles in three consecutive certification audits. Platinum facilities must successfully pass every audit without corrective action or observation and maintain continuous certification with no gaps between certification periods. Certification is valid for 2 years.
Yellow
Gold certification is the standard level of WRAP certification, awarded to facilities that demonstrate full compliance with WRAP’s 12 Principles. Certification is valid for 1 year.
Silver
WRAP may issue, or a facility may request, a Silver certificate if an audit finds it in substantial compliance with WRAP’s 12 Principles, but identifies minor non-compliances in policies, procedures or training must be addressed. Certification is valid for 6 months.
4. Kim Sora was honored to receive WRAP Certification
To meet export requirements to major markets around the world such as the US, Europe, etc. Kim Sora has made great efforts to achieve WRAP certification, demonstrating compliance with social responsibility and management standards. quality of Kim Sora factory.
Kim Sora’s garment products are produced according to a closed process and quality management according to Japanese standards, ensuring that all products have the same quality. As a manufacturer operating in the international market, the main export markets are Japan, Taiwan, Korea, Switzerland, America…. we hope to bring consumers products that meet standards. Quality standards according to “Japanese standards – European style – Affordable prices”.
LEARN MEDICAL MASKS THAT MEET JAPANESE STANDARDS JIS T 9001: BPV>99 – TOP QUALITY STANDARDS
Japan is one of the leading countries in many fields, especially industry. Japan’s set of industrial standards is also extremely strict and prestigious in the world, ensuring the highest product quality. That’s why products that meet Japanese standards are always trusted.
Japanese medical masks that meet JIS T 9001 standards are manufactured and packaged according to Japan’s top quality standards, ensuring safety and effectiveness in preventing the spread of infectious diseases.
Kim Sora is proud to bring customers medical mask products that meet Japanese production standards JIS T 9001 and serve the Japanese market.
Let’s find out in detail with Kim Sora what a medical mask that meets Japanese standard JIS T 9001 is.
What is Japan’s JIS T 9001 standard?
JIS standards are Japanese industrial standards, abbreviated from Japanese Industrial Standards. This is the standard system applied to all Japanese industrial activities. The procedures of this standard were developed by the Japan Industrial Standards Committee and issued by the Japan Standards Federation.
The scope of JIS’s participation is very diverse, from heavy industry to light industry, especially the field of steel, stainless steel and other production materials. In addition, household products such as soap and detergent also have their own JIS standard evaluation system.
JIS T standards are used in the medical device industry and safety applications.
Why should you use JIS T 9001 Medical Mask – Medical mask that meets Japanese quality standards.
JIS T 9001 medical mask ensures safety and effectiveness in preventing the spread of infectious diseases. Manufactured and packaged according to Japan’s top quality standards, the JIS T 9001 medical mask uses safe materials, does not irritate the skin and ensures good filtration and antibacterial properties.
Furthermore, the JIS T 9001 medical mask has the ability to filter dust and microscopic dust particles such as PM2.5, dust, and air pollution, helping to protect your health in polluted environments.
Kim Sora medical masks meet high quality standards
According to JIS T 9001 standards, to be assessed as meeting Japanese export medical mask standards, there must be a rigorous combination of many different criteria such as:
BFE (Bacterial Filtration Efficiency): Bacterial filtration efficiency
PFE (Particle Filtration Efficiency): particle filtration efficiency with a test particle size of 0.1 micrometers
VFE (Viral Filtration Efficiency): viral filtration efficiency
Along with three other test criteria:
Flammability: test the flammability of product materials
Blood barrier: blood barrier
Healthy and safety-breathabitity: safety and health criteria – product breathability.
Because of the rigor in evaluating such testing criteria, when buying Japanese medical masks – masks meeting JIS T 9001 standards, you can be assured of their ability to prevent the spread of infectious diseases. , protect the health of yourself and others.
Kim Sora medical masks are proud to meet Japanese export medical mask standards according to JIS T 9001 Class II standards, are popular with the Japanese market and are a supplier of JIS T 9001 medical masks in Vietnam. Male.
Currently, Kim Sora medical masks have been distributed nationwide. Please contact us to buy genuine products at the most preferential prices.
Why do you need to use standard medical masks and how to choose the right one?
In the article below, we will provide information about a type of European quality standard and introduce to you a type of medical mask that meets the recommended standard for use, which is a type of medical mask that meets European EN14683 standards. Europe.
The main purpose of medical masks is to protect patients from infectious agents. Additionally, in certain situations to protect the wearer against potentially contaminated liquids. Therefore, medical masks need to be selected from reputable suppliers that meet standards and ensure quality.
In addition, medical masks exported to demanding markets such as Europe must meet strict prescribed standards.
Kim Sora is proud to be a manufacturer and exporter of medical masks that meet EN 14683 Type II, Type IIR standards for the European market.
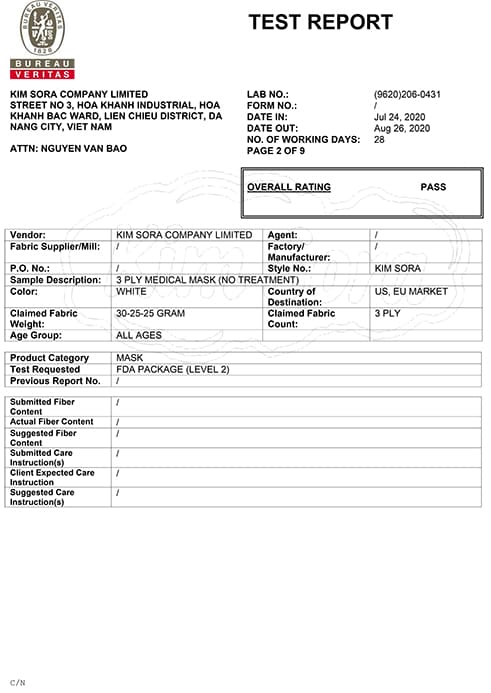
Learn about EN 14683 standard
EN14683 is a European standard that stipulates technical and testing requirements for exported medical mask products. This standard classifies medical mask products into three types (Type I, Type II, and Type IIR) based on their protective ability and level of air permeability.
Type I: This is a type of medical mask that is capable of blocking larger particles, but not capable of blocking smaller particles (like viruses). Type I masks are commonly used in hospitals to protect against large particles.
Type II: This type of medical mask is capable of blocking larger particles and smaller particles (such as viruses), with a lower level of air permeability than Type I masks. This type of mask is used in hospitals, clinics, and other places where the risk of infection is higher.
Type IIR: This type of medical mask is capable of blocking larger particles and smaller particles (like viruses), with a lower level of air permeability than type IIR masks. This type of mask is used in hospitals and other places where the risk of infection is higher. In addition, type IIR masks also have the ability to protect against spray droplets when speaking or sneezing from the person wearing the mask, helping to reduce the risk of spreading the disease to those around them.
Why should you use medical masks that meet European standard EN 14683?
EN14683 is a European standard that specifies technical and testing requirements for medical mask products, including requirements for filtration, air permeability and adhesion. EN14683 sets out detailed technical requirements for medical masks, including:
Filtration performance: Medical masks must be able to filter at least 98% of dust particles ranging in size from 0.1 to 5 micrometers.
Ability to barrier liquids: Medical masks must be able to prevent liquids, such as saliva or blood, from entering the mask.
Permeability: Medical masks must be able to prevent outside liquids from penetrating and coming into contact with the user’s skin.
Breathability: Medical masks must have enough ventilation for the user to breathe comfortably for a long time.
Antibacterial ability: Medical masks must be able to prevent bacteria and viruses.
Durability and adhesion: Medical masks must have enough durability and adhesion to stay firmly on the user’s face for a long time.
Design and size: Medical masks must have the appropriate design and size to fit the user’s face.
How to choose a standard mask
To meet the strict standards of the European market and the desire to bring products to Europe, Kim Sora has made efforts to develop products and test them to meet EN14683 standards: TYPE II, TYPE IIR. We are confident is the leading supplier and distributor of medical masks that meet export standards in the country.
In addition, Kim Sora medical masks are certified:
ISO 9001:2015: is a standard system for product quality management; recognized worldwide. Any business whose products meet the standards set by ISO; will be granted ISO 9001 certificate. When obtaining ISO 9001 certificate; proving that the enterprise has an effective quality management system and meets common international standards.
ISO 13485:2016 – is a standard for management systems applied in the field of manufacturing and trading of tools; medical supplies.
FDA: is to monitor and evaluate food quality; Whether the pharmaceutical product meets the criteria for import into the US?
CE: is a document indicating that the product has met health protection standards; safety and environmental protection according to European Union (EU) law and will be circulated in the European Economic Area (EEA).
As a manufacturer operating in the international market, the main export markets are Japan, Taiwan, Korea, Switzerland, America…. we hope to bring consumers products that meet standards. Quality standards according to “Japanese standards – European style – Affordable prices”.
Besides exporting medical masks, Kim Sora brand distributes and wholesales medical masks nationwide with price policy and commitment.
Top quality reputation nationwide.
With quality, standard products and reasonable prices, Kim Sora is a reliable address and trusted by many customers.
Please contact Kim Sora immediately for advice and to order today.
Latest update of Kim Sora’s ISO 13485:2016 certificate
Kim Sora is honored to continue to be evaluated and in accordance with the requirements of international standard ISO 13485:2016 for the field of production and distribution of medical masks issued by the Quality Testing and Certification Center (TQC). ). In the article below, we would like to provide some information related to the international certificate ISO 13485: 2016.
What is ISO 13485:2016 standard?
The latest version is ISO 13485:2016, a set of international standards for management systems applied in the field of manufacturing and trading of tools; medical supplies. Applied to ensure the ability to provide products that meet customer requirements and legal regulations.

How important is ISO 13485:2016?
The company’s medical equipment products benefit greatly from the input stage to the production process, including a quality management system certified to ISO 13485 standards. Below are a few factors that show its importance. The importance and benefits of ISO 13485 when businesses apply:
Provide safe products for users
Create competitive advantage, enhance brand, can be easily exported
Save costs, increase revenue and profits, minimize risks
Activities are managed systematically, helping to control product quality and safety
Improve production efficiency and product quality
Control hazards, control hygiene and contamination and have a specific implementation plan
Labor productivity increases
Improve the ability to meet customer requirements. Meet national and international regulations for medical products
Improve competitiveness for businesses and expand markets
Improve the efficiency of the current management system. Convenient integration with other management systems (ISO 9001, ISO/IEC 17025, ISO 14001).
ISO 13485:2016 – Latest version
Standard on Safety Management System for Medical Products is part of the ISO 13485:2003 standard set, the first version of which was issued by the international standardization organization ISO in July 2003. (Equivalent to the International Standard (TCVN ISO 13485:2004) ISO 13485:2012 (or BS EN ISO 13485:2012).
ISO 13485:2016 – The latest version was released to replace the old version ISO 13485:2003. This standard was published on 1 March 2016. There is a 3-year transition period from the old version to the new version for manufacturers and other organizations. That is until February 28, 2019 when ISO 13485:2003 expires. It also means that updating to the new version from the old version ISO 13485:2003 and the related European standard: EN ISO 13485:2012 needs to be done no later than this date.
Kim Sora is assessed with international certificate ISO 13485:2016
Continuing the quality standards evaluated in previous years, Kim Sora Co., Ltd. is honored to continue to be evaluated and in accordance with the requirements of international standard ISO 13485:2016 effective from September 25. March 2023 to March 24, 2026. This demonstrates the development and continuous improvement of product quality and production processes to bring quality – safe – effective products to Kim Sora’s customers.
In addition, Kim Sora medical masks are certified:

ASTM F2100-19 Level 1,2,3: is a standard of the American Institute of Standards and Technology (ASTM International) for medical masks used in medical procedures and infection prevention.
ISO 9001:2015: is a standard system for product quality management;
FDA: is to monitor and evaluate food quality; Whether the pharmaceutical product meets the criteria for import into the US?
CE: is a document indicating that the product has met health protection standards; safety and environmental protection in accordance with European Union (EU) legislation and will be marketed within the European Economic Area (EEA).
Please contact Kim Sora immediately for advice and to order today.
Email: sales@kimsora.com.vn
Hotline: 0905 986 797
Fanpage: Kim Sora Mask
I/ ASTM F2100-19 for the US market
For products belonging to the medical equipment group, specifically medical masks, gas masks, epidemic prevention kits… that want to be exported to the markets of developed countries such as the US and EU, it is necessary to ensure Ensure standard certificates comply with regulations.
Kim Sora is proud to be a manufacturer of medical masks that meets FDA certification and ASTM F2100-19 Level 2, Level 3 standards and exports to the US market.
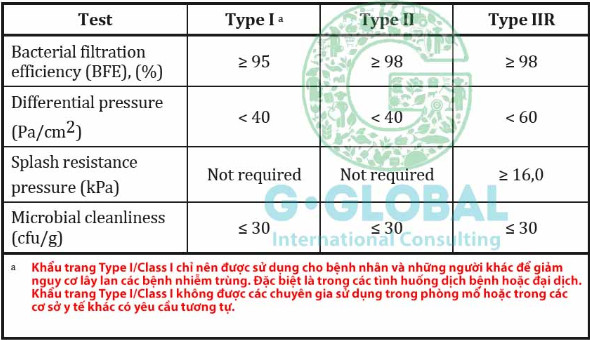
II/ Overview of ASTM F2100 standard
ASTM F2100 is a standard of the American Institute of Standards and Technology (ASTM International) for medical masks used in medical procedures and infection prevention.
The levels of the ASTM F2100 standard are divided into three levels, evaluating the filtering and protective ability of masks as follows:
Level 1: This is the lowest level in the ASTM F2100 standard, with the ability to filter at least 95% of particles larger than or equal to 0.1 microns. Level 1 masks are often used in situations where there is frequent contact with patients who are not infected with viruses or bacteria.
Level 2: This level is capable of filtering at least 98% of particles greater than or equal to 0.1 microns, and also provides protection for users against the risk of exposure to patients infected with viruses or bacteria. Level 2 masks are commonly used in hospitals, clinics, laboratories and other medical situations.
Level 3: This is the highest level in the ASTM F2100 standard, with the ability to filter at least 98% of particles greater than or equal to 0.1 microns and provide the highest protection for users. Level 3 masks are often used in special medical situations such as surgery and dangerous infectious diseases.
III/ Kim Sora medical masks meet export quality to the US market:
To meet the strict standards of the US market and the desire to bring products to the US, Kim Sora has made efforts to develop products and test them to meet ASTM F2100-19 Level 2, level 3 standards.


In addition, Kim Sora medical masks are certified:
ISO 9001:2015: is a standard system for product quality management; recognized worldwide. Any business whose products meet the standards set by ISO; will be granted ISO 9001 certificate. When obtaining ISO 9001 certificate; proving that the enterprise has an effective quality management system and meets common international standards.
ISO 13485:2016 – is a standard for management systems applied in the field of manufacturing and trading of tools; medical supplies.
FDA: is to monitor and evaluate food quality; Whether the pharmaceutical product meets the criteria for import into the US?
CE: is a document indicating that the product has met health protection standards; safety and environmental protection according to European Union (EU) law and will be circulated in the European Economic Area (EEA).
As a manufacturer operating in the international market, the main export markets are Japan, Taiwan, Korea, Switzerland, America…. we hope to bring consumers quality products. Quality according to “Japanese standards – European style – Affordable prices”.
In addition to export, the Kim Sora brand distributes and wholesales medical masks nationwide with a price policy and commitment to leading prestigious quality nationwide.
With quality products and reasonable prices, Kim Sora is a reliable address and trusted by many customers. Please contact this distributor for advice and to order today.
Learn more about Kim Sora masks__________











 Tiếng Việt
Tiếng Việt 日本語
日本語