4 LAYER MEDICAL FACE MASK OMEGA SEMI-CIRCLE
50.000 ₫
- Protect health.
- Prevent bacteria; Viruses and ultrafine dust help protect the respiratory tract when exposed to the outside environment.
- Especially safe for users, no skin irritation
Product name: 4 layers Medical Face mask Omega semi-circle

1.The Kim Sora Mask obtained the following certification:
- ISO 9001: 2015 – is the standard system for product quality management recognized worldwide. Any business whose products meet ISO’s set standards will be granted ISO 9001 certification. Upon obtaining ISO 9001 certification, it proves that the company has an effective and responsive quality management system. general international standards.
- ISO 13485: 2016 – is the standard of management system applied in the field of manufacturing – trading medical equipment and materials.
- FDA: is to monitor and evaluate the quality of food and pharmaceutical products in accordance with import criteria into the US or not.
- CE: is a type of document indicating that the product has met the standards for health, safety and environmental protection under the laws of the European Union (EU) and will be circulated in the European Economic Area. Europe (EEA).
Kim Sora mask products are proud of being a reputable brand, quality and health safety for consumers. For products consumed domestically or exported, we all test and filter criteria that are certified by international organizations. To ensure and certify that, you should review the following criteria test:
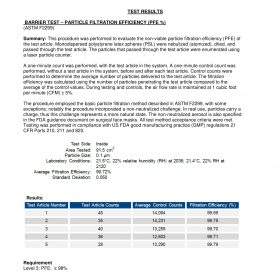
FILTER 3 CRITERIA:
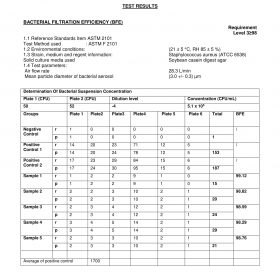
BFE> 99
PFE> 99
VFE> 99
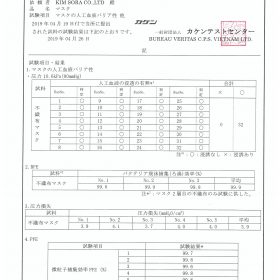
2. The test report is the test results issued by the testing institution as requested
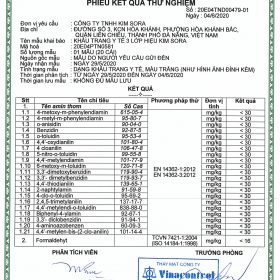
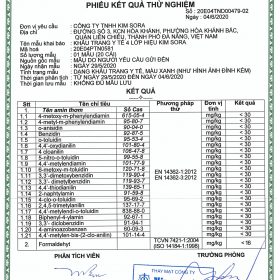
3. Test result sheet – Kim Sora 3-class medical mask
4. Test result card – Kim Sora 4-class medical mask
5. Test result sheet – Kim Sora antibacterial 4-layer medical mask
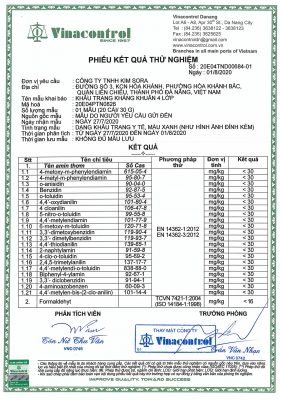

6. Test report of 4 grades reaching level III
7. Kaken
Be the first to review “4 LAYER MEDICAL FACE MASK OMEGA SEMI-CIRCLE” Cancel reply
You must be logged in to post a review.
Related products
Face mask
Face mask
Face mask
Face mask
Face mask
Face mask
Face mask
Face mask
Face mask
Face mask


 Tiếng Việt
Tiếng Việt 日本語
日本語

















































Reviews
There are no reviews yet.